Indications, Important Safety Information, and Prescribing Information. BOTOX® Cosmetic (onabotulinumtoxinA) Important Information Indications BOTOX® Cosmetic (onabotulinumtoxinA) is indicated in adult patients for the temporary improvement in the appearance of: – Moderate to severe glabellar lines associated with corrugator and/or procerus muscle activity – Moderate to severe lateral canthal lines associated with orbicularis oculi activity – Moderate to severe forehead lines associated with frontalis activity IMPORTANT SAFETY INFORMATION, INCLUDING BOXED WARNING WARNING: DISTANT SPREAD OF TOXIN EFFECT Postmarketing reports indicate that the effects of BOTOX® Cosmetic and all botulinum toxin products may spread from the area of injection to produce symptoms consistent with botulinum toxin effects. These may include asthenia, generalized muscle weakness, diplopia, ptosis, dysphagia, dysphonia, dysarthria, urinary incontinence, and breathing difficulties. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life threatening and there have been reports of death. The risk of symptoms is probably greatest in children treated for spasticity, but symptoms can also occur in adults treated for spasticity and other conditions, particularly in those patients who have an underlying condition that would predispose them to these symptoms. In unapproved uses and approved indications, cases of spread of effect have been reported at doses comparable to those used to treat cervical dystonia and spasticity and at lower doses. CONTRAINDICATIONS BOTOX® Cosmetic is contraindicated in the presence of infection at the proposed injection site(s) and in individuals with known hypersensitivity to any botulinum toxin preparation or to any of the components in the formulation. WARNINGS AND PRECAUTIONS Lack of Interchangeability Between Botulinum Toxin Products The potency units of BOTOX® Cosmetic are specific to the preparation and assay method utilized. They are not interchangeable with other preparations of botulinum toxin products and, therefore, units of biological activity of BOTOX® Cosmetic cannot be compared to nor converted into units of any other botulinum toxin products assessed with any other specific assay method. Spread of Toxin Effect Please refer to Boxed Warning for Distant Spread of Toxin Effect. No definitive serious adverse event reports of distant spread of toxin effect associated with dermatologic use of BOTOX® Cosmetic at the labeled dose of 20 Units (for glabellar lines), 24 Units (for lateral canthal lines), 40 Units (for forehead lines with glabellar lines), 44 Units (for simultaneous treatment of lateral canthal lines and glabellar lines), and 64 Units (for simultaneous treatment of lateral canthal lines, glabellar lines, and forehead lines) have been reported. Patients or caregivers should be advised to seek immediate medical care if swallowing, speech, or respiratory disorders occur. Serious Adverse Reactions With Unapproved Use Serious adverse reactions, including excessive weakness, dysphagia, and aspiration pneumonia, with some adverse reactions associated with fatal outcomes, have been reported in patients who received BOTOX® injections for unapproved uses. In these cases, the adverse reactions were not necessarily related to distant spread of toxin, but may have resulted from the administration of BOTOX® to the site of injection and/or adjacent structures. In several of the cases, patients had pre-existing dysphagia or other significant disabilities. There is insufficient information to identify factors associated with an increased risk for adverse reactions associated with the unapproved uses of BOTOX®. The safety and effectiveness of BOTOX® for unapproved uses have not been established. Hypersensitivity Reactions Serious and/or immediate hypersensitivity reactions have been reported. These reactions include anaphylaxis, serum sickness, urticaria, soft-tissue edema, and dyspnea. If such reactions occur, further injection of BOTOX® Cosmetic should be discontinued and appropriate medical therapy immediately instituted. One fatal case of anaphylaxis has been reported in which lidocaine was used as the diluent and, consequently, the causal agent cannot be reliably determined. Cardiovascular System There have been reports following administration of BOTOX® of adverse events involving the cardiovascular system, including arrhythmia and myocardial infarction, some with fatal outcomes. Some of these patients had risk factors including pre-existing cardiovascular disease. Use caution when administering to patients with pre-existing cardiovascular disease. Increased Risk of Clinically Significant Effects With Pre-existing Neuromuscular Disorders Individuals with peripheral motor neuropathic diseases, amyotrophic lateral sclerosis, or neuromuscular junction disorders (eg, myasthenia gravis or Lambert-Eaton syndrome) should be monitored when given botulinum toxin. Patients with neuromuscular disorders may be at increased risk of clinically significant effects including generalized muscle weakness, diplopia, ptosis, dysphonia, dysarthria, severe dysphagia, and respiratory compromise from onabotulinumtoxinA (see Warnings and Precautions). Dysphagia and Breathing Difficulties Treatment with BOTOX® and other botulinum toxin products can result in swallowing or breathing difficulties. Patients with pre-existing swallowing or breathing difficulties may be more susceptible to these complications. In most cases, this is a consequence of weakening of muscles in the area of injection that are involved in breathing or oropharyngeal muscles that control swallowing or breathing (see Boxed Warning). Pre-existing Conditions at the Injection Site Caution should be used when BOTOX® Cosmetic treatment is used in the presence of inflammation at the proposed injection site(s) or when excessive weakness or atrophy is present in the target muscle(s). Dry Eye in Patients Treated With BOTOX® Cosmetic There have been reports of dry eye associated with BOTOX® Cosmetic injection in or near the orbicularis oculi muscle. If symptoms of dry eye (eg, eye irritation, photophobia, or visual changes) persist, consider referring patients to an ophthalmologist. Human Albumin and Transmission of Viral Diseases This product contains albumin, a derivative of human blood. Based on effective donor screening and product manufacturing processes, it carries an extremely remote risk for transmission of viral diseases and variant Creutzfeldt-Jakob disease (vCJD). There is a theoretical risk for transmission of Creutzfeldt-Jakob disease (CJD), but if that risk actually exists, the risk of transmission would also be considered extremely remote. No cases of transmission of viral diseases, CJD or vCJD have ever been identified for licensed albumin or albumin contained in other licensed products. ADVERSE REACTIONS The most frequently reported adverse reactions following injection of BOTOX® Cosmetic for glabellar lines were eyelid ptosis (3%), facial pain (1%), facial paresis (1%), and muscular weakness (1%). The most frequently reported adverse reaction following injection of BOTOX® Cosmetic for lateral canthal lines was eyelid edema (1%). The most frequently reported adverse reactions following injection of BOTOX® Cosmetic for forehead lines with glabellar lines were headache (9%), brow ptosis (2%), and eyelid ptosis (2%). DRUG INTERACTIONS Co-administration of BOTOX® Cosmetic and aminoglycosides or other agents interfering with neuromuscular transmission (eg, curare-like compounds) should only be performed with caution as the effect of the toxin may be potentiated. Use of anticholinergic drugs after administration of BOTOX® Cosmetic may potentiate systemic anticholinergic effects. The effect of administering different botulinum neurotoxin products at the same time or within several months of each other is unknown. Excessive neuromuscular weakness may be exacerbated by administration of another botulinum toxin prior to the resolution of the effects of a previously administered botulinum toxin. Excessive weakness may also be exaggerated by administration of a muscle relaxant before or after administration of BOTOX® Cosmetic. USE IN SPECIFIC POPULATIONS There are no studies or adequate data from postmarketing surveillance on the developmental risk associated with use of BOTOX® Cosmetic in pregnant women. There are no data on the presence of BOTOX® Cosmetic in human or animal milk, the effects on the breastfed child, or the effects on milk production. Please see BOTOX® Cosmetic full Prescribing Information including Boxed Warning and Medication Guide.
Important Information INDICATIONS JUVÉDERM® VOLUX™ XC injectable gel is indicated for subcutaneous and/or supraperiosteal injection for improvement of jawline definition in adults over the age of 21 with moderate to severe loss of jawline definition. JUVÉDERM® VOLUMA® XC injectable gel is indicated for deep (subcutaneous and/or supraperiosteal) injection for cheek augmentation to correct age-related volume deficit in the mid-face and for augmentation of the chin region to improve the chin profile in adults over the age of 21. IMPORTANT SAFETY INFORMATION CONTRAINDICATIONS These products should not be used in patients who have severe allergies, marked by a history of anaphylaxis or history or presence of multiple severe allergies, and should not be used in patients with a history of allergies to Gram-positive bacterial proteins or lidocaine contained in these products. WARNINGS Do not inject into blood vessels. Introduction of these products into the vasculature may lead to embolization, occlusion of the vessels, ischemia, or infarction. Take extra care when injecting soft tissue fillers. For example, after insertion of the needle and just before injection, the plunger rod can be withdrawn slightly to aspirate and verify the needle is not intravascular. Inject the product slowly, and apply the least amount of pressure necessary. Rare, but serious, adverse events associated with the intravascular injection of soft tissue fillers in the face have been reported and include temporary or permanent vision impairment, blindness, cerebral ischemia or cerebral hemorrhage leading to stroke, skin necrosis, and damage to underlying facial structures. Immediately stop the injection if a patient exhibits any of the following symptoms: changes in vision, signs of a stroke, blanching of the skin, or unusual pain during or shortly after the procedure. Patients should receive prompt medical attention and, possibly, evaluation by an appropriate healthcare professional specialist should an intravascular injection occur Product use at specific sites in which an active inflammatory process (skin eruptions such as cysts, pimples, rashes, or hives) or infection is present should be deferred until the underlying process has been controlled PRECAUTIONS To minimize the risk of potential complications, these products should only be used by healthcare professionals who are knowledgeable about the anatomy and the product(s) for use in indicated area(s), and who have appropriate training in facial anatomy, vasculature, safe injection techniques, and identification and management of potential adverse events, including intravascular complications The potential risks of soft tissue injections should be discussed with patients prior to treatment to ensure they are aware of signs and symptoms of complications The safety and effectiveness for the treatment of anatomic regions other than indicated areas for each product have not been established in controlled clinical studies The safety for use of these products in patients with known susceptibility to keloid formation, hypertrophic scarring, and pigmentation disorders has not been studied The safety for use of JUVÉDERM® VOLUMA® XC has been established in patients between 35 and 65 years of age for cheek augmentation and in patients between 22 and 80 years of age for chin augmentation. The safety for use of JUVÉDERM® VOLUX™ XC in patients under 22 years has not been established The safety for use during pregnancy and in breastfeeding females has not been established Dermal filler implantation carries a risk of infection. Follow standard precautions Dermal fillers should be used with caution in patients on immunosuppressive therapy Patients taking medications that can prolong bleeding (such as aspirin, nonsteroidal anti-inflammatory drugs, and warfarin) may experience increased bruising or bleeding at treatment sites If laser treatment, chemical peel, or any other procedure based on active dermal response is considered after treatment, or before skin has healed from a procedure prior to treatment, there is a possible risk of eliciting an inflammatory reaction at the injection site The safety for use of JUVÉDERM® VOLUMA® XC injectable gel in patients with very thin skin in the mid-face has not been established The safety for use of JUVÉDERM® VOLUMA® XC with cannula for cheek augmentation has not been established in patients with Fitzpatrick Skin Types V and VI JUVÉDERM® VOLUMA® XC was not evaluated in subjects with significant skin laxity of the chin, neck, or jaw in the chin augmentation study The effect of JUVÉDERM® VOLUMA® XC injection into the chin on facial hair growth has not been studied Patients may experience late-onset adverse events with use of injectable gel implants, and late-onset nodules with use of JUVÉDERM® VOLUMA® XC Based on preclinical studies, patients should be limited to 20 mL of any JUVÉDERM® injectable gel per 60 kg (132 lb) body mass per year. The safety of injecting greater amounts has not been established Injection of more than 9 mL of JUVÉDERM® VOLUX™ XC for improvement of jawline definition has not been studied ADVERSE EVENTS The most common reported side effects for JUVÉDERM® injectable gels were redness, swelling, pain, tenderness, firmness, lumps/bumps, bruising, discoloration, and itching. The majority were mild or moderate in severity. To report an adverse reaction with any product in the JUVÉDERM® Collection, please call Allergan® Product Support at 1-877-345-5372. Please visit JuvedermDFU.com for more information. Products in the JUVÉDERM® Collection are available only by a licensed physician or properly licensed practitioner.
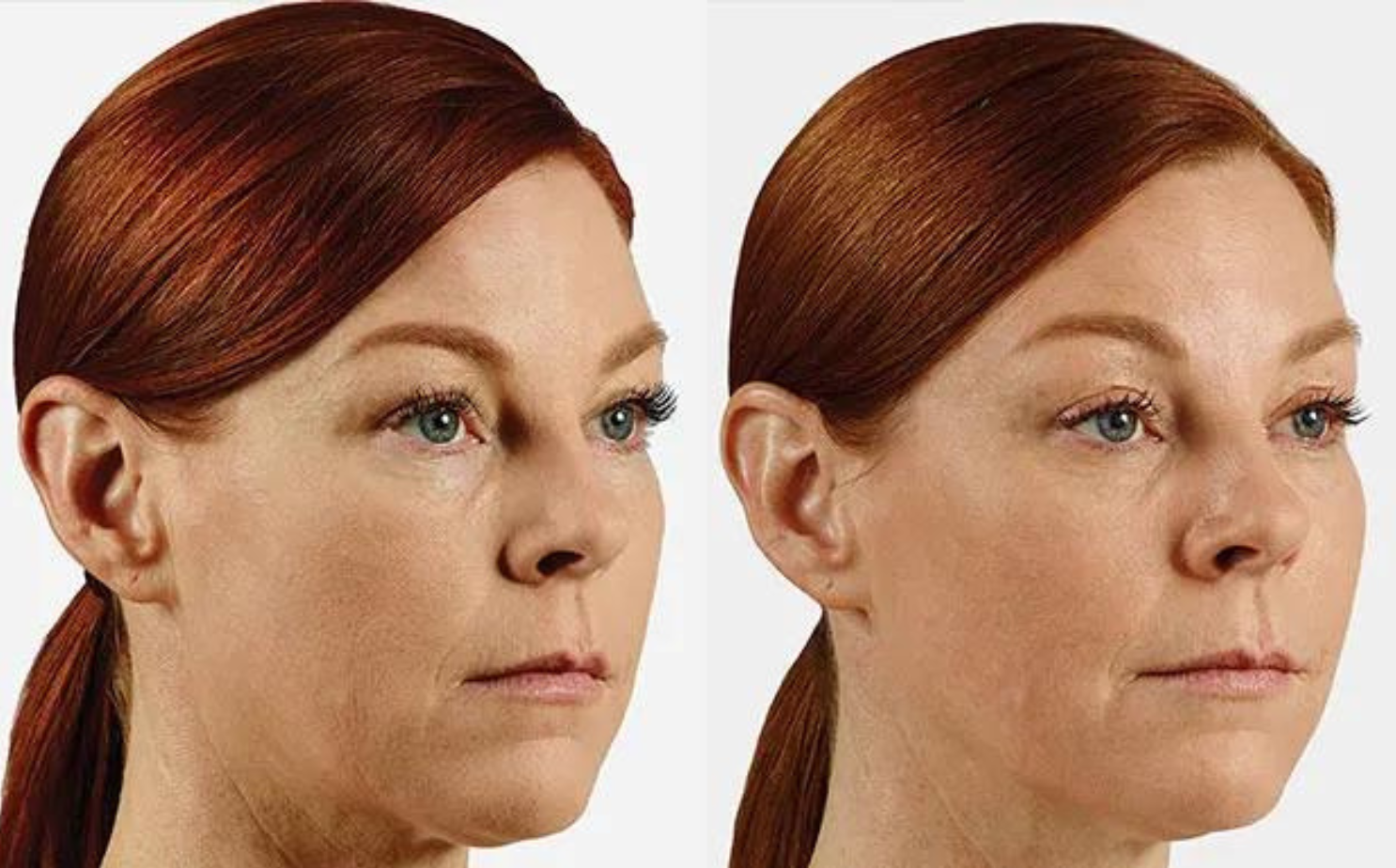
BOTOX Treatment for Fine Lines and Wrinkles
At Milwaukee Laser & Body Aesthetics, we offer Botox treatments for your face and hyaluronic acid fillers as effective solutions for reducing wrinkles and enhancing facial features. Botox temporarily relaxes muscles to smooth dynamic wrinkles, while fillers add volume to areas with static wrinkles or volume loss. Our skilled practitioners utilize a tasteful amount of fillers/injectables to prevent overdoing, ensuring natural-looking results tailored to your unique needs and preferences. Both treatments can be used together for comprehensive facial rejuvenation, which can also be paired with our scar reduction treatments for an all-encompassing approach to skin care. Consulting with a qualified practitioner can help determine the best approach for achieving desired results.
| Purpose | Smooths crow’s feet and forehead wrinkles |
| Appointment length | 45 minutes |
| Time between appointments | 3-4 months |
| Downtime | None |
| Payment plan eligible? | Yes, through Cherry |
BOTOX Treatment
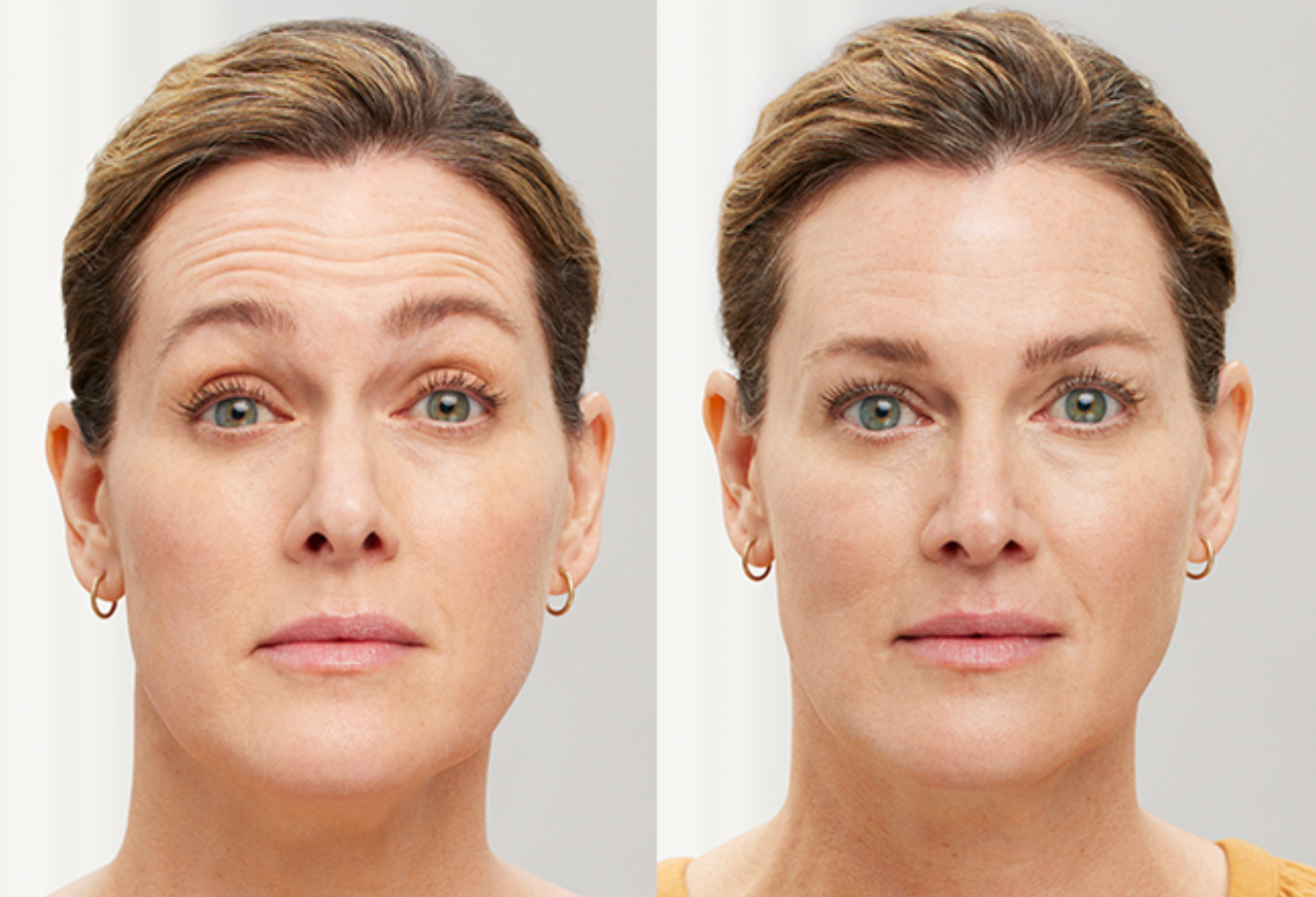
BOTOX, also known as botulinum toxin, is a popular cosmetic treatment used to reduce the appearance of wrinkles and fine lines on the face. BOTOX works by temporarily relaxing the muscles causing wrinkles to smooth the skin and giving it a more youthful appearance. When injected into specific muscles, BOTOX blocks the release of acetylcholine, a neurotransmitter responsible for muscle contraction. By inhibiting muscle activity, BOTOX prevents the formation or deepening of wrinkles caused by repetitive facial expressions, such as frown lines, crow's feet and forehead wrinkles.
Our patients find several aspects of BOTOX treatments to be appealing:
- The swift nature of the procedure allows for minimal disruption to their daily routines, with most treatments taking 45 minutes.
- Noticeable transformations occur rapidly, typically within 10-14 days.
- Our approach to BOTOX treatments offers a high degree of customization. The price and aggressiveness of the treatment can be tailored to individual needs and preferences.
What BOTOX Can Improve
BOTOX works well for adults looking to soften visible lines caused by repeated facial movements. Treatment smooths common expression areas like crow’s feet, forehead lines and furrowed brows. Some clients also use BOTOX proactively to delay the development of deeper creases. Our practitioners customize every session for natural-looking, balanced results without affecting your unique features.
How Much Does Treatment Cost?
At Milwaukee Laser & Body Aesthetics, we offer personalized pricing for Botox and fillers to match the uniqueness of each individual. Our treatment plans and pricing are customized to your specific needs. During a complimentary, 30-minute consultation, our Clinic Manager will assess the area, skin type and other relevant factors to determine the cost of treatment. Due to this highly tailored approach, we cannot provide quotes over the phone or without an in-person consultation.
We offer pricing for both individual treatment sessions and package options. MLBA also offers financing plans for eligible treatments.
Juvéderm® Treatment for Augmentation
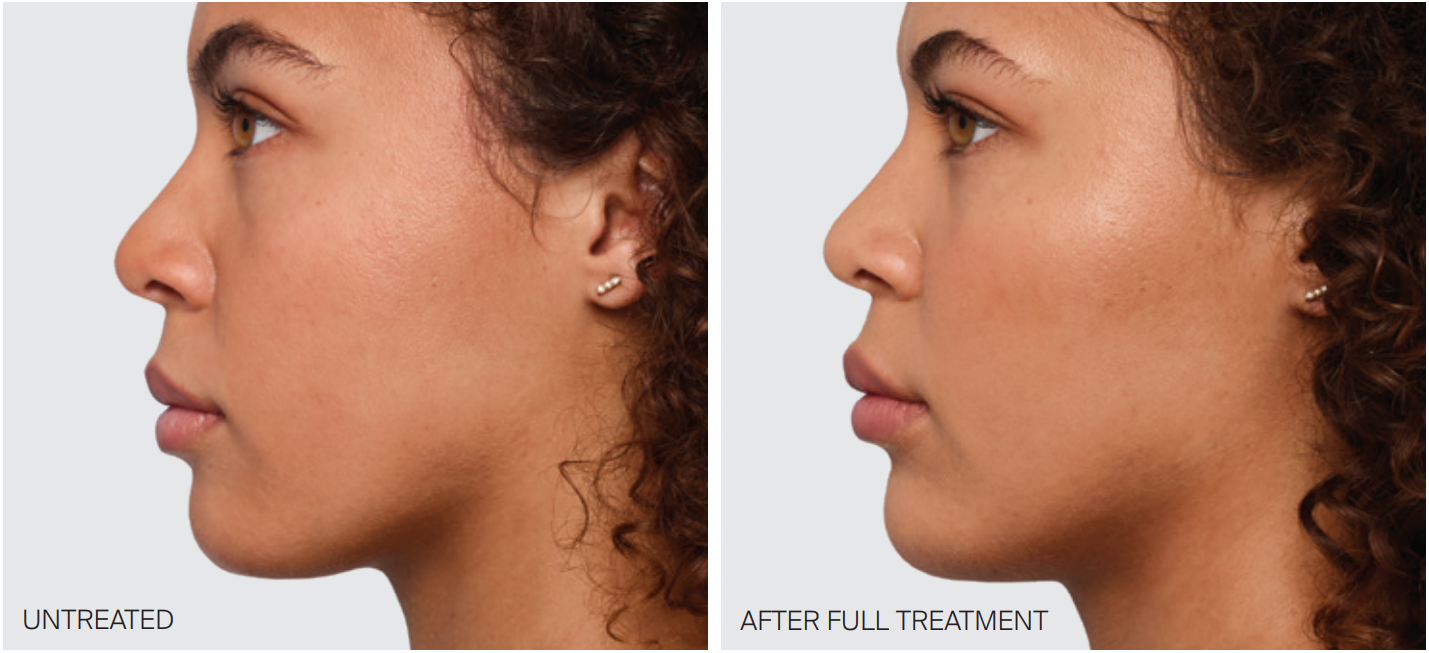
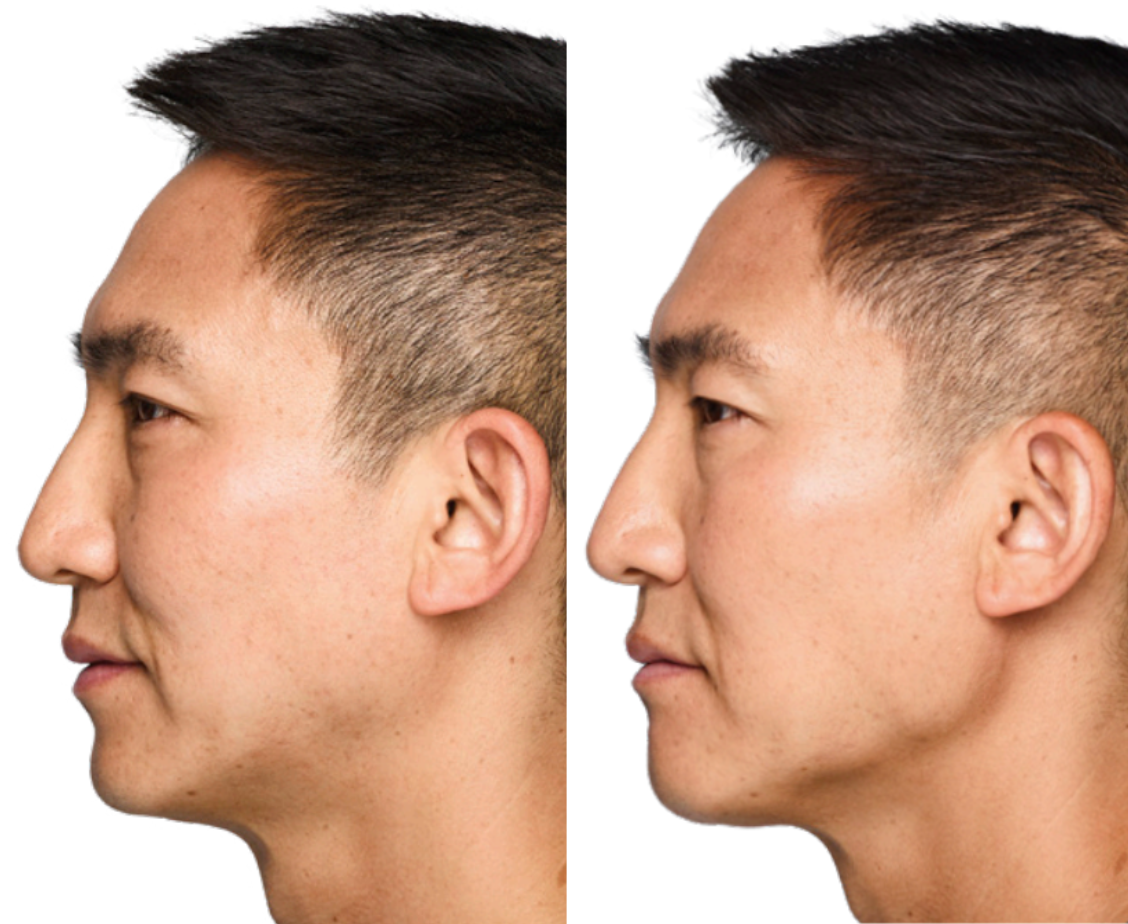
Juvéderm is a brand of hyaluronic acid-based dermal fillers commonly used for facial rejuvenation and the correction of wrinkles and volume loss in the skin. Hyaluronic acid occurs naturally in the body and helps maintain moisture and volume in the skin. When injected, Juvéderm fillers add volume, smooth fine lines and enhance facial contours, providing a temporary yet effective solution for reducing signs of aging. Milwaukee Laser & Body Aesthetics utilizes the VOLBELLA®, VOLLURE® and VOLUMA® injectables from Allergan Aesthetics’ JUVÉDERM product line.
| Purpose | For facial rejuvenation, lip augmentation, wrinkle reduction (e.g., nasolabial folds), cheek augmentation or chin augmentation |
| Appointment length | 30 - 60 minutes |
| Time between appointments | Up to 18 months |
| Downtime | None |
| Payment plan eligible? | Yes, through Cherry |
Hyaluronic Acid Treatment for Plumping
Are you looking for an anti-aging treatment with instant results? Nothing works as quickly as hyaluronic acid fillers, offering instant results like our advanced hair removal treatments. Hyaluronic acid fillers assist in lip plumping, elevating cheekbones, eliminating wrinkles, defining the jawline and much more. Sessions typically last 1 hour and are done as needed, with results lasting longer than standard injections like BOTOX. There is no downtime and results are instantaneous.
| Purpose | Plumps lips, elevates cheekbones, reduces wrinkles, defines the jawline & more |
| Appointment length | 1 hour |
| Time between appointments | Up to 18 months |
| Downtime | None |
| Payment plan eligible? | Yes, through Cherry |


.jpg)
.jpg)
.jpg)
.jpg)
.jpg)



